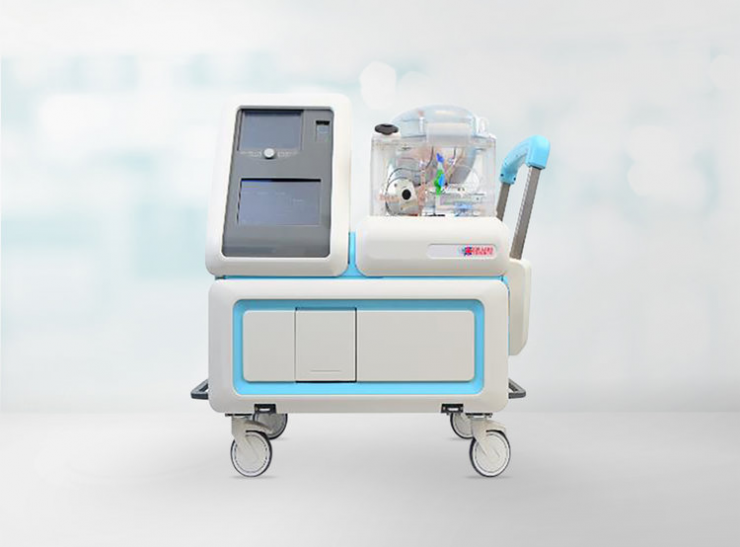
Closely-held XOR Labs hopes to dramatically increase the number of lungs suitable for transplantation with its Ex-Vivo Lung Perfusion (EVLP) technology.
“Only about 20 out of every 100 donated lungs are currently suitable for transplant worldwide, but our aim is to boost this to 60% with our EVLP platform,” Dr. Thomas Waddell, CEO, says in an interview with BioTuesdays.
Dr. Waddell contends that XOR’s EVLP technology has the potential to help hundreds of thousands of people who are in critical need for lung transplantation due to diseases such as pulmonary fibrosis, cystic fibrosis, pulmonary hypertension and chronic obstructive pulmonary disease.
“Our ultimate goal is to reduce waiting list deaths to zero and to become the worldwide leader in advanced multi-organ transplantation technology,” he adds.
According to industry estimates, more than 100,000 people would benefit from a lung transplant, but only 6,000 are performed annually. And some 20% to 30% of patients die while waiting from a transplant. “The key limiting factor is insufficient quality donor lungs available for transplantation,” he suggests.
“Only about 20 out of every 100 donated lungs are currently suitable for transplant worldwide, but our aim is to boost this to 60% with our EVLP platform”
Dr. Waddell participated in the scientific and clinical development of the world renowned “Toronto EVLP protocol,” led by Dr. Shaf Keshavjee, used at University Health Network (UHN), which allows for re-evaluation, repair and eventually regeneration of donated lungs. He and his team also have led the effort to translate the Toronto protocol into a XOR EVLP platform.
“The Toronto EVLP is the gold standard of care for lung transplantation and has been used about 400 times over the past nine years, resulting in some 300 transplants,” he points out, In addition, the Toronto protocol has increased utilization of donated lungs to 40%. “Our aim is to move the utilization needle higher still.”
The EVLP process keeps lungs nourished and alive outside the body for up to 12 hours by circulating blood-free fluid through the lungs at body temperature, while the lungs are gently ventilated.
This gives lungs, which would have been discarded, time to be assessed, and transplanted, if they perform well, Dr. Waddell says. In addition, injured lungs can be rehabilitated instead of discarded, and if sufficiently recovered, can be transplanted.
Dr. Waddell explains that Dr. Keshavjee and a team of leading thoracic surgeons and researchers from UHN, who helped develop the original Toronto system, founded XOR Labs with the goal of commercializing a standardized and mobile version of the Toronto system for transplant patients around the world. The company hopes to unlock the pool of potential donors and expand lung transplants to 15,000 to 20,000 a year, from 6,000 currently.
“Our system simplifies the EVLP process and drastically cuts hospital costs for lung transplants,” he contends. In addition, its “mobility makes it uniquely suited for out-of-hospital use, opening up opportunities to use the EVLP process during the transportation of donated organs.”
The XOR EVLP System is comprised of a re-usable cart, which includes perfusion pumps, sensors and a ventilator; a single-use Lung Tower where the lung is placed; and several per-procedure sterile single-use cannulas to connect the donor lungs to the Lung Tower.
“In animal models, Dr. Keshavjee’s research team has kept lungs going for 24 hours, which allows us to contemplate some interesting therapy options,” Dr. Waddell says.
XOR hopes to begin a three-month first-in-man validation trial of its EVLP device as early as January 2019 in order to seek Health Canada approval. The trial will be conducted at Toronto General Hospital with 25 EVLP patients and will require 20 transplants. “We hope the Canadian data would allow us to submit an application for CE Mark approval in Europe,” he adds.
The company is seeking an additional investment of up to $7-million to complete Health Canada and CE Mark approvals and begin its entry into the U.S. market.
In Europe, XOR has partnered with Xenios, a member of the Fresenius Healthcare Group, for manufacturing, regulatory, infrastructure, and sales and marketing support. Xenios also owns a minority interest in XOR, which has already had discussions with key clinical leaders in China, Europe, and the U.S.
Dr. Waddell says discussions also have begun with the FDA to establish an “early feasibility evaluation pathway” for its technology that would lead to a humanitarian device exemption or premarket approval application.
Dr. Keshavjee and Dr. Waddell have also established a close relationship with United Therapeutics, which is leading the introduction of EVLP into the U.S., using the “Toronto protocol” and hopes to sell XOR devices into their “service centers” as well.
“Our five-year plan includes installing 36 XOR ELVP carts in 25 transplant or perfusion centers around the world,” he adds.
• • • • •
To connect with XOR, or any of the other companies featured on BioTuesdays, send us an email at [email protected].